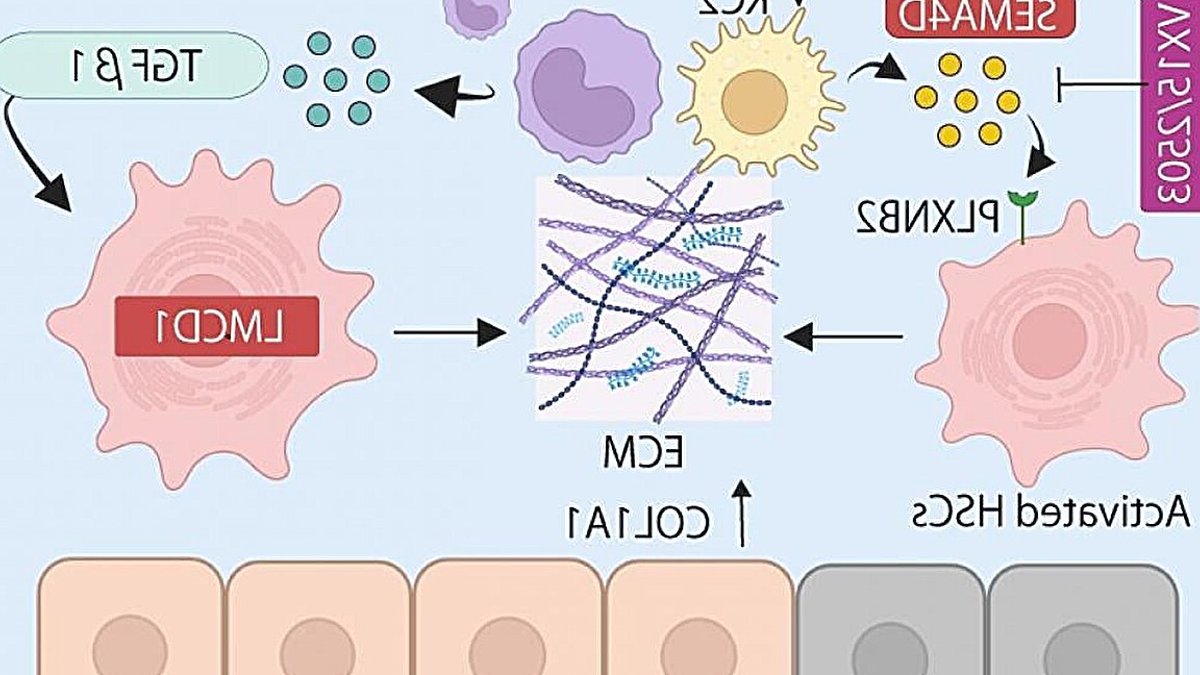
Liver fibrosis, a progressive hardening and scarring of liver tissue, can advance to cirrhosis and liver cancer if left untreated. Osaka Metropolitan University researchers have identified two proteins, semaphorin-4D (SEMA4D) and LIM and cysteine-rich domains 1 (LMCD1), that drive fibrosis and can be blocked.
These proteins were discovered using a cutting-edge technique called single-cell fixed RNA profiling, which analyzes the gene activity of individual liver cells. The researchers applied this technique to liver cells from healthy, fibrotic, and recovering mice.
Read Also: Endoscopic procedure outperforms semaglutide in short-term weight loss
The study found that SEMA4D acts as a “distress signal” that binds to a receptor on hepatic stellate cells, activating them and increasing collagen production. LMCD1, on the other hand, operates as an internal “master switch” that keeps stellate cells in an active, scar-producing state.
Blocking SEMA4D with a monoclonal antibody or silencing LMCD1 reduced fibrosis in mice. The researchers also found that both proteins correlated with fibrosis severity in human samples.
The discovery of these two proteins could lead to the development of a combination antifibrotic therapy that attacks fibrosis from multiple angles. This therapy could potentially reduce the progression of the disease and lower the risk of liver cancer.
Associate Professor Le Thi Thanh Thuy, the co-corresponding author of the study, said, “The fact that SEMA4D and LMCD1 work through completely different mechanisms and in two different cell types—macrophages and stellate cells—is what excites us most.”
The researchers hope that their work will inform the development of the first genuinely effective antifibrotic therapies. The goal is not just to slow scarring down, but to actively stop it and push the liver toward recovery.
The study’s findings were published in the journal JHEP Reports. The researchers used a technique that produces a precise picture of each individual cell’s behavior, even from frozen tissue samples.
This technique, called single-cell fixed RNA profiling, allowed the researchers to analyze the gene activity of approximately 38,000 individual liver cells. The result was a detailed map of how every major liver cell type changes as fibrosis progresses and retreats.
The researchers found that hepatocytes in the pericentral zone, the central region of each liver lobule, show a striking restoration of their normal gene activity during recovery. This suggests that this specific hepatocyte population plays an active role in driving liver regeneration.
The discovery of SEMA4D and LMCD1 as key drivers of liver fibrosis has significant implications for the treatment of the disease. By targeting both these proteins using a combination therapy, patients could potentially reduce the progression of the disease.
According to the researchers, the difference between patients who heal and those who do not lies in whether their livers can successfully shut down these pro-fibrotic signals. By identifying the precise proteins and cell interactions that tip this balance, the team hopes their work will inform the development of effective antifibrotic therapies.
The study’s findings are based on research conducted in mice and human samples. The researchers used a humanized monoclonal antibody to block SEMA4D and silencing LMCD1 to reduce fibrosis in mice.
The researchers also analyzed human liver biopsy samples and found that both these proteins correlated with fibrosis severity. The levels of SEMA4D and LMCD1 declined in patients who cleared hepatitis C, which correlated with a reduced risk of liver cancer progression.
The study’s results suggest that targeting SEMA4D and LMCD1 could be a promising approach to treating liver fibrosis. The researchers hope that their work will lead to the development of effective therapies for this devastating disease, and they will be able to develop a combination therapy that attacks fibrosis from multiple angles, using these two proteins as targets.




Leave a Reply