Schizophrenia, a serious brain disorder, affects about 23 million people worldwide, with cognitive dysfunction present in over 80% of patients. It causes confused thinking, severe memory problems, and hallucinations.
A research group led by scientists from Nagoya University in Japan has tested a drug used to treat an immune disease to see if it could reduce schizophrenia-related symptoms in mice.
Read Also: Your Ultimate Guide to Recovering Quickly from Inguinal Hernia Surgery
The findings, published in Molecular Psychiatry, show that KD025 restored connections between neurons and significantly improved memory and visual recognition in mice, without causing the serious side effects common to current schizophrenia medications.
Current medications help with some symptoms, but they often do not improve cognitive function. They also cause serious side effects such as hormonal disruptions, involuntary muscle movements, and weight gain, which leads to many patients stopping treatment.
Researchers focused on a gene called ARHGAP10. Variants of this gene are much more common in people with schizophrenia than in the general population.
“ARHGAP10 controls the activity of a brain protein called ROCK2. In mice with these genetic variants, ROCK2 becomes overactive. In a previous study, we found that this overactivity appears to damage connections between neurons and impair cognition,” said Rinako Tanaka, co-lead author and former project assistant professor at Nagoya University’s Graduate School of Medicine.
The team tested KD025, approved in the United States to treat an immune disease called chronic graft-versus-host disease, which can occur after bone marrow transplants.
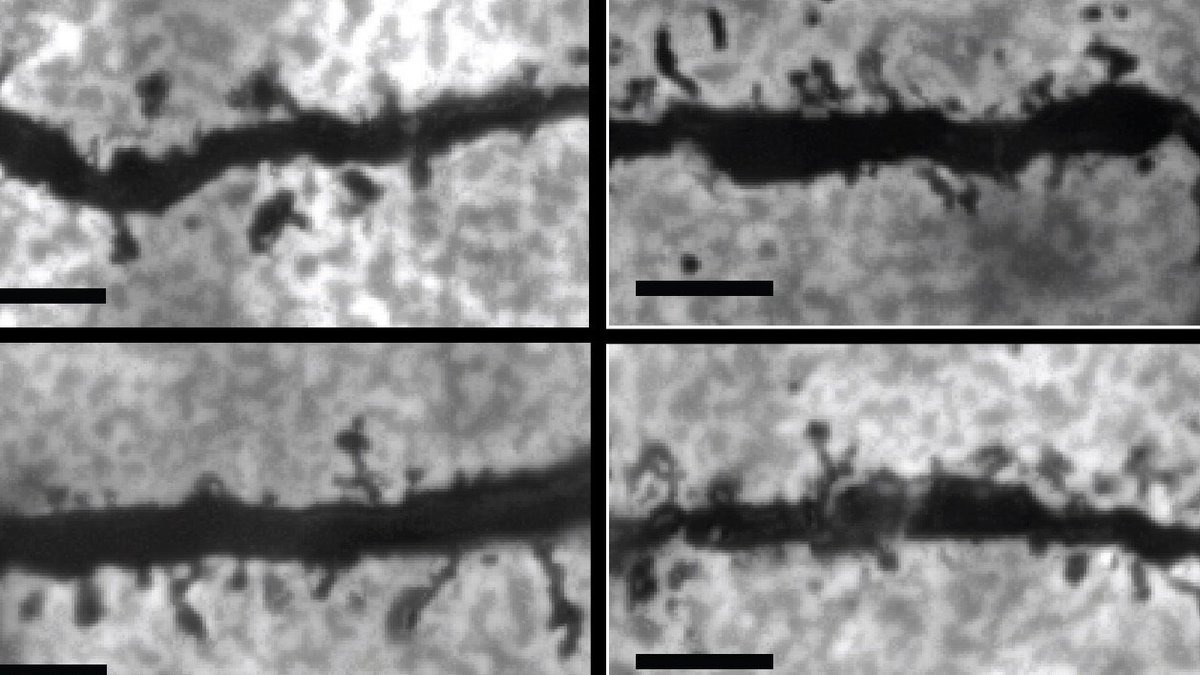
In mice engineered to carry schizophrenia-associated gene variants, the drug decreased the overactivity of ROCK2. KD025 also restored the density of tiny structures on neurons called dendritic spines, which are critical for memory.
These had been reduced in mice carrying schizophrenia-associated gene variants. The drug had no effect on healthy mice.
“Importantly, KD025 did not cause the side effects typical of current antipsychotic drugs. At effective doses, it caused no involuntary movements, hormonal abnormalities, or significant changes in blood pressure or blood sugar,” said Hiroyuki Mizoguchi, co-author and associate professor from the Department of Neuropsychopharmacology and Hospital Pharmacy at Nagoya University.
Because KD025 has already been through clinical safety trials for another condition, human trials for schizophrenia could start sooner than for a new drug.
While the researchers caution that all experiments were in mice, and human studies are needed, the findings point to a promising target for treatments that are more effective and better tolerated by patients.
Future studies will investigate how KD025 improves brain cell connections and function, and further evaluate its safety and efficacy to support human trials.
In a room on the third floor of the university, the researchers spent countless hours analyzing data and discussing their findings.
The study’s results are published in the journal Molecular Psychiatry, with a DOI of 10.1038/s41380-026-03567-7.
The Nagoya University team’s work was done between 9 am and 5 pm, Monday through Friday, over the course of several months.
For now, the researchers are waiting for the green light to start human trials, which could potentially lead to a new treatment for schizophrenia.
According to the report, KD025 may offer a safer and more effective treatment option for cognitive dysfunction in schizophrenia, pending human trials.




Leave a Reply